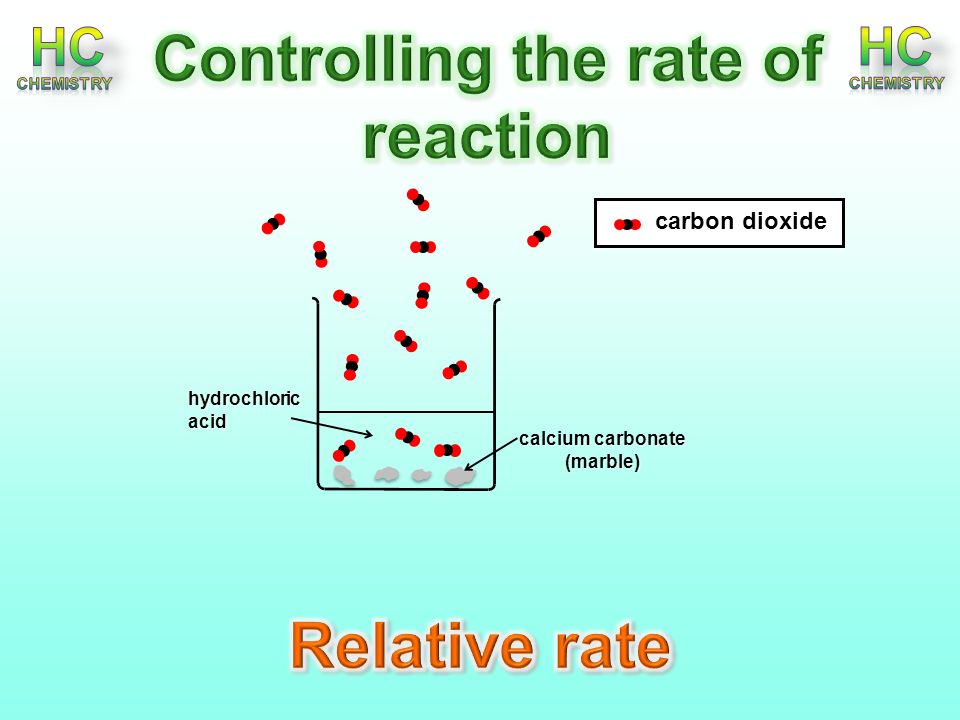
A Piece Of Marble Reacts With 2 00 L

A piece of marble assume it is pure caco 3 reacts with 2 00 l of 2 52 m hc1 after dissolution of the marble a 10 00 ml sample of the resulting solution is withdrawn added to some water and titrated with 24 87 ml of 0 9987 m naoh.
A piece of marble reacts with 2 00 l. The percentage of c a c o 3 in the marble is. First you measure out 25cm3 of hydrochloric acid 2. Secondly weight out 3 grams of marble chips for each concentration 3. After dissolution of the marble a 10 00.
After dissolution of the marble a 10 ml sample of the remaining hcl aq is withdrawn added to some water and titrated with 24 87 ml of 9987 m naoh. Blue color p 8. Pour the acid into the conical flask and add the marble chips 5. You then fill a bowl with water along with a boiling tube and straight after attach the delivery tube at the end of the boiling tube 4.
Determined without changing matter reacts with something else. A piece of marble assume it to be pure caco3 reacts with 2 00 l of 2 52 m hcl. After dissolution of the marble a 10 00 ml sample of the remaining hcl is withdrawn added to some water and titrated with 24 87 ml of 0 9987 m naoh. Answer to a piece of marble assume it is pure caco3 reacts with 2 00 l of 2 52 m hc1.
It is composed primarily of the mineral calcite caco 3 and usually contains other minerals such as clay minerals micas quartz pyrite iron oxides and graphite under the conditions of metamorphism the calcite in the limestone recrystallizes to form a rock that is. Matter will be changed into a new. After dissolution of the marble a 10 00 ml sample of the remaining hcl aq is withdrawn added to. When the reaction was complete 1 1 2 0 c m 3 of o 2 was obtained s t p.
Substance after the reaction. Identify the following as a chemical c or physical property p. What was the mass of the peice of marble. Marble is a metamorphic rock that forms when limestone is subjected to the heat and pressure of metamorphism.
Lapis lazuli artifacts. A piece of marble assume it to be pure caco 3 reacts with 2 00 l of 2 52 m hcl. 10 gram of a piece of marble was put into excess of dilute hcl acid. What must have been the mass of the piece of marble.